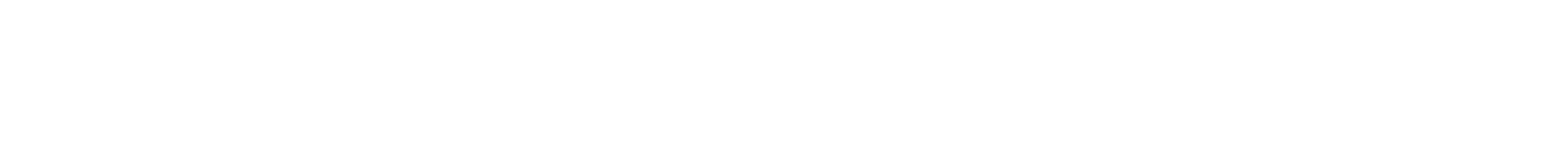Chiral amines as initiators for ROP and chiral induction on poly(2-aminoisobutyric acid) chains
Rohmer, M.. et. al. Polymer Chemistry, 2021, 12, 6252-6262, DOI: 10.1039/D1PY01021B
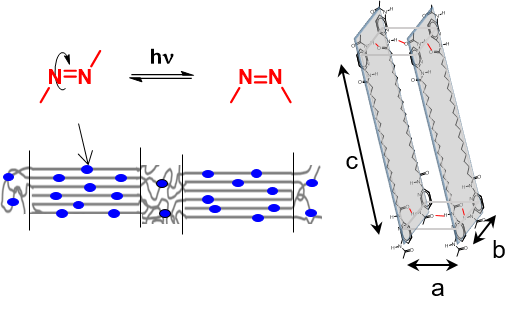
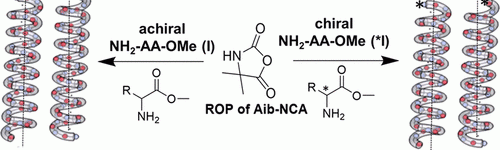
2-Aminoisobutyric acid (Aib) is a prominent achiral amino acid known for its helical building properties, generating left- and right-handed helices without any preference. We here report on several short chain poly(Aib)s synthesized by ring opening polymerization (ROP) of Aib-N-carboxy-anhydrides (Aib-NCA) with structurally different chiral amines acting as both, initiator and chiral induction agent. By adding a chiral center at the C-terminus of the polymer chain we can control the direction of the screw sense, since the polymers are adopting the chirality of the initiator following the sergeant-and-soldier principle. Variation of the solvent from hexafluoroisoropanol (HFIP) to water resulted in a significant change of CD signals, featuring a surprising aggregation of the poly(amino acids) into globular aggregates in water in the range of 50 to 200 nm, which accounts for changes in chiral induction. Published with a permission of the Polymer Chemistry 2021.