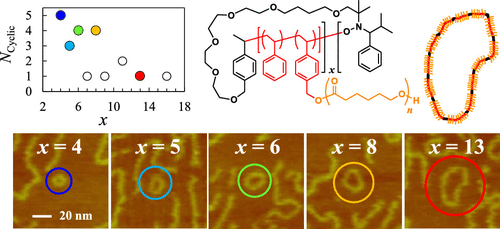
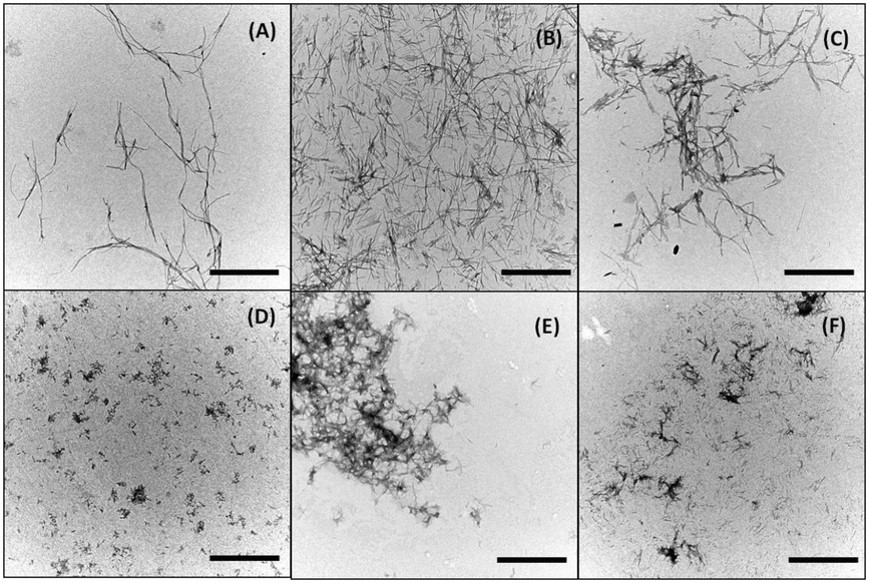
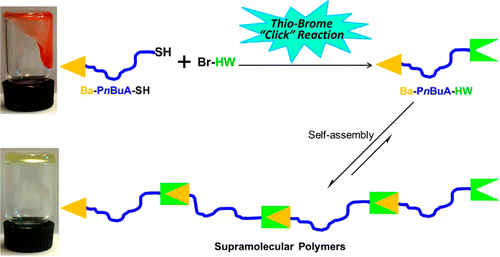
Döhler, D., et al. Accounts of Chemical Research 2017, 50 (10), 2610-2620, DOI: http://dx.doi.org/10.1021/acs.accounts.7b00371.
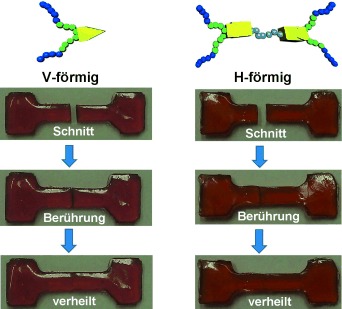
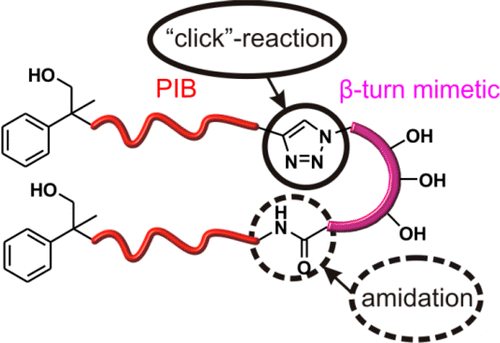
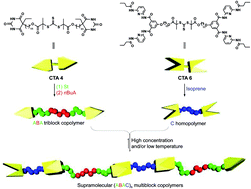
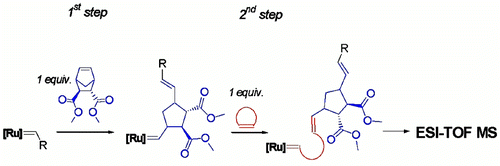
Click chemistry has emerged as a significant tool for materials science, organic chemistry, and bioscience. The use of the CuAAC in self-healing systems, most of all the careful design of copper-based catalysts linked to additives as well as the chemical diversity of substrates, has led to an enormous potential of applications of this singular reaction. The examples reported here describe chemical concepts to realize more efficient and faster click reactions in self-healing polymeric materials, via enhanced chain diffusion in (hyper)branched polymers, autocatalysis, or internal chelation concepts enable efficient click cross-linking already at 5 °C. We have designed special CuAAC click methods for chemical reporting and visualization systems based on the detection of ruptured capsules via a fluorogenic click reaction and we have prepared polymeric Cu(I)–biscarbene complexes to detect (mechanical) stress within self-healing polymeric materials via a triggered fluorogenic reaction, thus using a destructive force for a constructive chemical response. Reproduced with permission. Copyright 2017©, American Chemical Society.