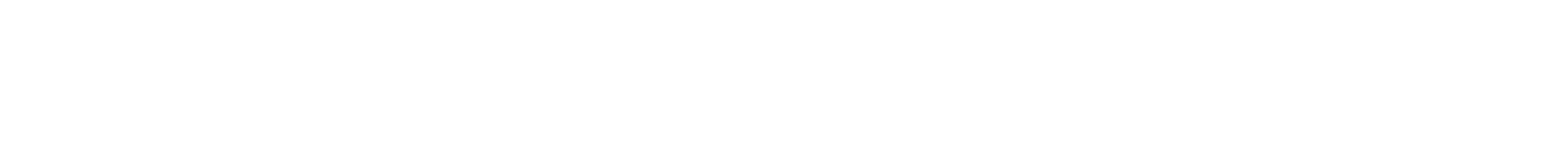Fluorinated Linkers Enable High-Voltage Pyrrolidinium-based Dicationic Ionic Liquid Electrolytes
Katcharava, Z. et. al., Chemistry- An European Journal., 2024, 30, e202402004, https://doi.org/10.1002/chem.202402004
Novel fluorinated, pyrrolidinium-based dicationic ionic liquids (FDILs) as high-performance electrolytes in energy storage devices have been prepared, displaying unprecedented electrochemical stabilities (up to 7 V); thermal stability (up to 370 °C) and ion transport (up to 1.45 mS cm−1). FDILs were designed with a fluorinated ether linker and paired with TFSI/FSI counterions. To comprehensively assess the impact of the fluorinated spacer on their electrochemical, thermal, and physico-chemical properties, a comparison with their non-fluorinated counterparts was conducted. With a specific focus on their application as electrolytes in next-generation high-voltage lithium-ion batteries, the impact of the Li-salt on the characteristics of dicationic ILs was systematically evaluated. The incorporation of a fluorinated linker demonstrates significantly superior properties compared to their non-fluorinated counterparts, presenting a promising alternative towards next-generation high-voltage energy storage systems. © 2024 The Authors. Chemistry – A European Journal published by Wiley-VCH GmbH